Insulin Drug Interactions: Subtle but Dangerous
It is important for practitioners to understand traditional insulin dosing requirements and recognize special situations in which ther- apy modifications should be made.
More than 29.1 million Americans live with diabetes.1All patients with type 1 diabetes (T1D) and many with type 2 diabetes (T2D) require insulin therapy for adequate glucose control. Insulin is a natural hormone produced by pancreatic B cells that induces glucose uptake into peripheral tissues. Insulin does not interact with other medications through traditional cytochrome P450 enzyme mechanisms. However, it does require dosing adjustments in many instances.
It is important for practitioners to understand traditional insulin dosing requirements and recognize special situations in which therapy modifications should be made. Because of their unconventional nature, drug and disease state interactions with insulin may go unnoticed and cause severe patient harm.
CLINICAL GUIDELINES ON INSULIN DOSING
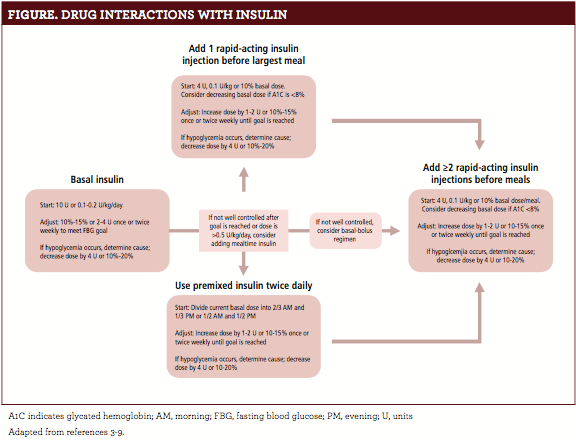
The American Diabetes Association (ADA) released updated guidelines in 2017 that include insulin dosing recommendations. Providers should consider starting insulin in symptomatic, newly diagnosed patients with high fasting blood glucose or glycated hemoglobin levels. Basal insulin (insulin glargine, insulin detemir, insulin degludec) doses begin at 10 units daily or 0.1 to 0.2 units/kg/day and are titrated to goal glucose levels.2Some patients may require bolus mealtime insulin to lower glucose effectively. The ADA recommends using rapid-acting (insulin lispro, insulin aspart) formulations because of their quick onset. Thefigure2outlines a standard approach to initiating insulin therapy in patients with T2D.
INSULIN AND CORTICOSTEROIDS
Glucocorticoids such as prednisone and dexamethasone are commonly prescribed to treat inflammation, allergic reactions, asthma, and many other conditions. Unfortunately, they also increase blood glucose levels by promoting insulin resistance via altered insulin signaling pathways in skeletal muscle, liver, and adipose tissue.3Glucocorticoids can also increase visceral adipose tissue by inducing the differentiation of preadipocytes to mature fat cells.
Patients with diabetes who are receiving steroids should frequently monitor their blood glucose levels. Insulin may require an adjustment depending on the dose, frequency, and length of glucocorticoid therapy. Guidelines recommend increasing all rapid- and short-acting insulins by 10%.4Basal insulin may need to be increased if fasting glucose readings rise. Providers must also remember to readjust the patient’s insulin when steroid treatment ends.
INFECTION
Acute illnesses can cause large blood sugar fluctuations in patients with diabetes. Infection induces a stress response by the body, prompting the release of glucagon, epinephrine, and cytokines.
These molecules act on peripheral tissues to decrease insulin resistance, increase glucose production, and suppress insulin secretion.5Their presence creates a markedly elevated hyperglycemic state. With increased blood glucose and additional mechanisms at work, glucose-lowering therapy must be adjusted in patients with diabetes who have infections. Increasing insulin by 10% to 20% of the patient’s usual dose is typically recommended.6Providers should also consider other factors such as the infection’s severity and the patient’s ability to maintain adequate oral intake.
Some antibiotics can also affect blood sugars. Fluoroquinolones such as levofloxacin and gatifloxacin stimulate insulin secretion and may cause hypoglycemia.7Case reports describe sulfa antibiotics like sulfamethoxazole/trimethoprim causing hypoglycemia. Consider additional monitoring and insulin dose reductions when using these medications.
ANTIDIABETIC AGENTS
Dozens of oral and injectable antidiabetic medications are available today. Despite different mechanisms of action, these medications can have synergistic effects with insulin, producing profound hypoglycemia in patients if dosed incorrectly. Hypoglycemia, a scary event, is associated with negative outcomes. Large-scale clinical trials have shown that patients with histories of 1 or more severe hypoglycemic events are 2 to 4 times more likely to die than well-controlled patients.2
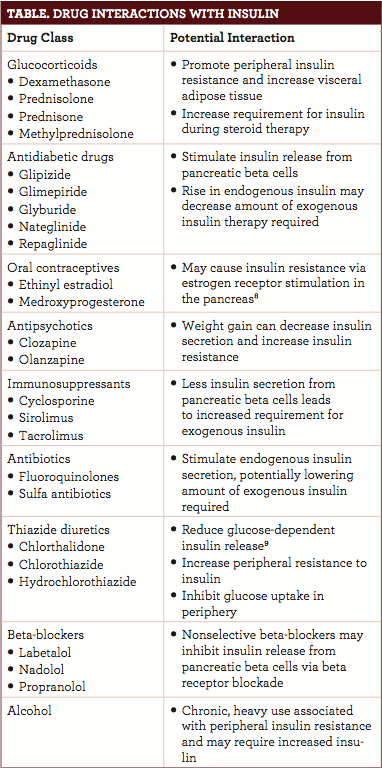
Sulfonylureas (glipizide, glimepiride, glyburide) have been widely implicated as causes of hypoglycemia. These medications directly stimulate insulin release from pancreatic beta cells. Nateglinide and repaglinide also increase insulin production. While these medications are commonly used in combination with insulin for patients who are unresponsive to single-agent therapy, the risk of hypoglycemia is additive. Providers must educate patients on combination therapy’s potential dangers, hypoglycemia’s warning signs, and what to do if hypoglycemia develops. Thetabledescribes medications that can potentially alter blood glucose and require insulin adjustments.
CONCLUSION
Insulin dosing is highly variable and largely patient specific. For the best patient outcomes, providers must be aware of insulin dosing protocols and recognize when adjustments should be made. Knowledge of a patient’s medications is an important step toward assuring safety and minimizing risk.
David Cabral, PharmD, is a pharmacist with Rite Aid Pharmacy in New Hampshire.
References
- American Diabetes Association. Statistics about diabetes. http://www.diabetes.org/diabetes-basics/statistics/. Updated 2016. Accessed March 20, 2017.
- American Diabetes Association. Promoting health and reducing disparities in populations. Sec. 1. In: Standards of Medical Care in Diabetes 2017.Diabetes Care. 2017;40(suppl 1):S6-S10.
- Ferris HA, Kahn CR. New mechanisms of glucocorticoid-induced insulin resistance: make no bones about it.J Clin Invest. 2012;122(11):3854-3857. doi: 10.1172/JCI66180.
- Lansang MC, Hustak LK. Glucocorticoid-induced diabetes and adrenal suppression: how to detect and manage them.Cleve Clin J Med. 2011;78(11):748-756. doi: 10.3949/ccjm.78a.10180.
- McCowen KC, Malhotra A, Bistrian BR. Stress-induced hyperglycemia.Crit Care Clin. 2001;17(1):107-124.
- Nau KC, Lorenzetti RC, Cucuzzella M, Devine T, Kline J. Glycemic control in hospitalized patients not in intensive care: beyond sliding-scale insulin.Am Fam Physician. 2010;81(9):1130-1135.
- Saraya A, Yokokura M, Gonoi T, Seino S. Effects of fluoroquinolones on insulin secretion and beta-cell ATP-sensitive K+ channels.Eur J Pharmacol.2004;497(1):111-117.
- Gourdy P. Diabetes and oral contraception.Best Pract Res Clin Endocrinol Metab.2013;27(1):67-76. doi:10.1016/j.beem.2012.11.001.
- Duarte JD, Cooper-DeHoff RM. Mechanisms for blood pressure lowering and metabolic effects of thiazide and thiazide-like diuretics.Expert Rev Cardiovasc Ther. 2010;8(6):793-802.

