Rx Product Updates (December 2015)
Read about the new Rx products featured in December.
Sanofi US and Regeneron Pharmaceuticals
Indication:The FDA has approved Praluent (alirocumab), a proprotein convertase subtilisin kexin type 9 inhibitor antibody, as adjunct to diet and maximally tolerated statin therapy for the treatment of adults with heterozygous familial hypercholesterolemia or clinical atherosclerotic cardiovascular disease who require additional lowering of low-density lipoprotein cholesterol (LDL-C). The recommended starting dose is 75 mg subcutaneously once every 2 weeks. If the LDL-C response is inadequate, the dosage may be increased to the maximum 150 mg every 2 weeks.
Dosage Form:Injection: 75 mg/mL or 150 mg/mL solution in a single-dose prefilled pen or syringe
For More Information:praluent.com
TUZISTRA XRMarketed by:Vernalis Therapeutics, Inc, and Tris Pharma, Inc
Indication:The FDA has approved Tuzistra XR—a combination of codeine, an opiate agonist antitussive, and chlorpheniramine, a histamine-1 receptor antagonist—for relief of cough and symptoms associated with upper respiratory allergies or the common cold. Tuzistra XR is not indicated for patients younger than 18 years. The recommended dose in adults 18 years and older is 10 mL every 12 hours, not to exceed 2 doses (20 mL) in 24 hours.
Dosage Form:Extended-release oral suspension: an antitussive, codeine polistirex, which contains 14.7 mg of codeine; and an antihistamine, chlorpheniramine polistirex, which contains 2.8 mg of chlorpheniramine per 5 mL
For More Information:vernalis.com/ development-2/commercial-pipeline/ coughcold/tuzistra-xr
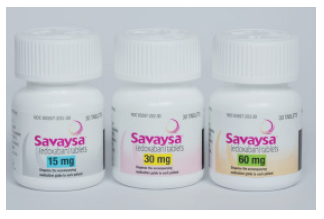
SAVAYSAMarketed by:Daiichi Sankyo, Inc
Indication:The FDA has approved Savaysa (edoxaban), a factor Xa inhibitor, to reduce the risk of stroke and systemic embolism in patients with nonvalvular atrial fibrillation. The recommended dose is 60 mg once daily in patients with a creatinine clearance (CrCl) >50 to ≤95 mL/min.; Savaysa should not be used in patients with a CrCl >95 mL/min. The dose should be reduced to 30 mg once daily in patients with a CrCl of 15 to 50 mL/ min. Savaysa already has FDA approval for the treatment of deep vein thrombosis and pulmonary embolism following 5 to 10 days of initial therapy with a parenteral anticoagulant.
Dosage Form:Tablets: 60, 30, and 15 mg
For More Information:savaysa.com

